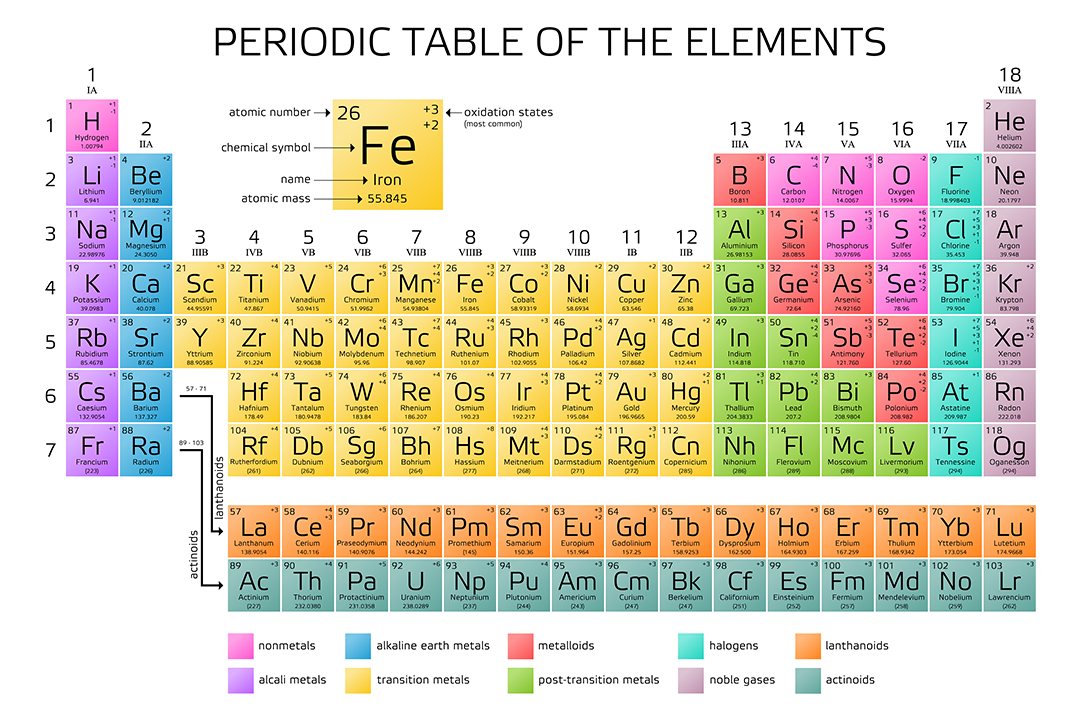
Eighteen columns divide the elements into groups with closely related physical properties. 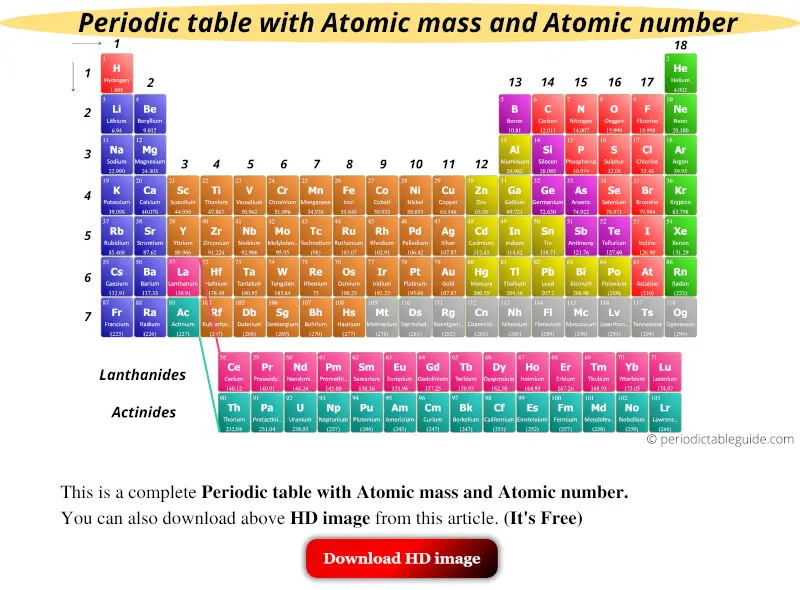
The periodic table arranges the elements, with their diverse physical and chemical properties, in order of atomic number and fits them into a logical pattern. You will need to refer to a periodic table for proton values. named elements that make up the periodic table (IUPAC 2016). In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. This color periodic table contains the accepted standard atomic weights (atomic masses) of. Lund (Anoka-Ramsey Community College), and Henry Agnew. This page is shared under a CK-12 license and was authored, remixed, and/or curated by Melissa Alviar-Agnew, Lance S. The relative atomic mass of an element is a weighted average of the masses of the atoms of the isotopes. 1: Moles to Mass Conversion with Elements. In addition to the elements symbol, most periodic tables will also contain the elements atomic number and the elements atomic mass. Mass number is simply the number of protons plus the number of neutrons in an atom when looking at a periodic table. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. Color Periodic Table of the Elements: Atomic Masses. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. Other periodic tables in this version of the text may refer to it as Ununbium (Uub).\nonumber \] †Element 112 named shortly before the release of this text. Legend of the IUPAC Periodic Table of the Elements and Isotopes 12 (prepared by and used with permission of Sara Glidewell). Options for hiding the symbol or name of the elements provide a handy learning. An uncertainty in the last digit in the Atomic Mass column is shown by the number in parentheses e.g., 1.00794(7) indicates ☐.00007. Chemistry Portal v t e The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows ('periods') and columns ('groups'). Use this periodic table for calculating molar mass for any chemical formula. Three such elements (Th, Pa, and U), however, do have a characteristic terrestrial isotopic composition, and an atomic mass is given for them. It is defined as the average mass of atoms of an element, which is calculated with the help of relative abundance of isotopes of a naturally occurring element. , indicates the mass number of the longest-lived isotope of the element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
